Voluntary Recall of Optase Intense Multidose
Scope Health Inc is conducting a voluntary retail level recall of 15 batches of Optase Dry Eye Intense Drops that were produced by a former manufacturing supplier and shipped to retail and eyecare professional customers between May 2024 and November 2025. The recall is being conducted out of an abundance of caution and in coordination with the U.S. Food and Drug Administration (FDA). Consumers/patients do not need to take any action. There have been no reports of serious adverse events associated with the impacted product.
Only the following lots listed in the table provided are impacted by this recall. The lot number is located on the top of the product carton where the red box appears in the example below:
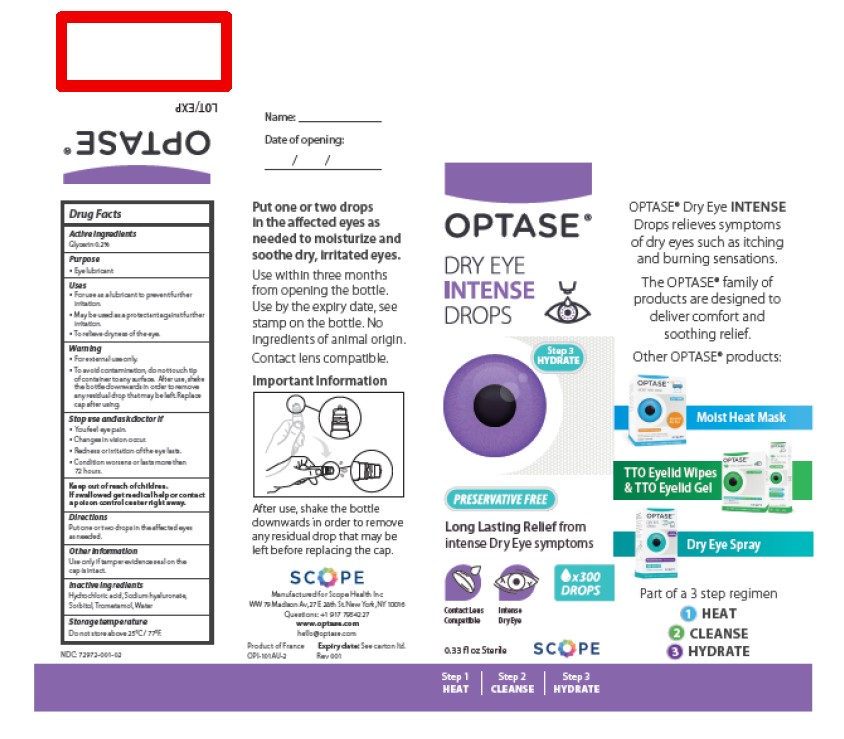
| Lot | Expiration Date* |
|---|---|
| 8T98, 9T31, 9T32 | 30/04/26 |
| 2V13, 2V14, 2V15 | 30/06/26 |
| 3V35 | 31/08/26 |
| 3V36, 3V37 | 30/09/26 |
| 5V45, 5V46, 9V12 | 31/03/27 |
| 1X57, 1X70, 1X84 | 31/05/27 |
* If you have a bottle with an expiration date of June 1st 2027 or after this is unaffected by the recall.
The recall is being conducted following the FDA’s recommendation based on the agency’s findings during a January 2026 inspection of Fareva Excelvision, the company that previously manufactured this product.
Scope Health Inc fully transitioned distribution of Optase Dry Eye Intense Drops to a new manufacturer by November 2025.
The recall will have no impact on product supply for customers or the patients who use the product.
Questions or Assistance?
If you require further details regarding this voluntary recall or need assistance with the next steps, our team is ready to support you. You can reach us through any of the following channels:
- Contact Form: Submit your inquiry directly here.
- Email Support: Message us at hello@optase.com.
- Customer Care Line: Call our dedicated team at +1 917 7954227.
We appreciate your cooperation and patience as we take these precautionary steps to ensure the quality of our products.







